Three Methods You can Eradicate Paxlovid Out Of Your Online Business
“At this time, Paxlovid is generally not recommended for these who're pregnant or nursing,” Dr, Jimmy Johannes, a pulmonologist and important care medication specialist at MemorialCare Long Seashore Medical Center in California, instructed Healthline. That concern can now be put aside because all the several real-world research confirming the protecting profit against hospitalizations and deaths are in people who've been vaccinated, and a significant proportion obtained booster pictures. The Food and Drug Administration (FDA) has authorized bivalent vaccine booster photographs from Pfizer-BioNTech for children and adults 5 years of age and older, and from Moderna for these 6 and older. The United States began rolling out the updated COVID pictures in September, designed to take on the circulating BA.Four and BA.5 Omicron subvariants and the unique version of the virus. The remedy is available by prescription for people and must be began inside 5 days of the onset of signs.
- Your Subsequent House Might be Made on an Assembly Line
- Heart disease
- Trouble swallowing or breathing
- Feeling generally unwell
- Pale-colored stools (poop)
In preregistration trials, Paxlovid began inside 5 days of symptom onset demonstrated a 89% reduction in subsequent hospitalizations for COVID-19 (1.0% vs 6.7%) and a major reduction in 28-day mortality (none vs 1.6%). Primarily based upon these outcomes and the continuing COVID-19 pandemic, Paxlovid was granted Emergency Use Authorization (EUA) in December 2021 as therapy of nonhospitalized patients (adults and children 12 years or older) with documented COVID-19 infection who're at excessive risk of complications. COVID-19 Deaths Since October 11, 2021: 84, 156 U.S. Merck submitted molnupiravir to the FDA to contemplate for emergency authorization on October 11, 2021-71 days in the past. NEW YORK, Sept 28 (Reuters) - Drugmaker Merck & Co mentioned on Wednesday that it agreed to permit China's Sinopharm to distribute and import its COVID-19 antiviral molnupiravir in China if the drug is authorised for use there. Bivalent means the shot protects against two strains of a virus, and these COVID-19 boosters are designed to protect towards both the original SARS-CoV-2 virus and the Omicron variant. Paxlovid is an inhibitor of CYP3A (Cytochrome P450, family 3, subfamily A) and may improve plasma concentrations of medication that are primarily metabolised by CYP3A.
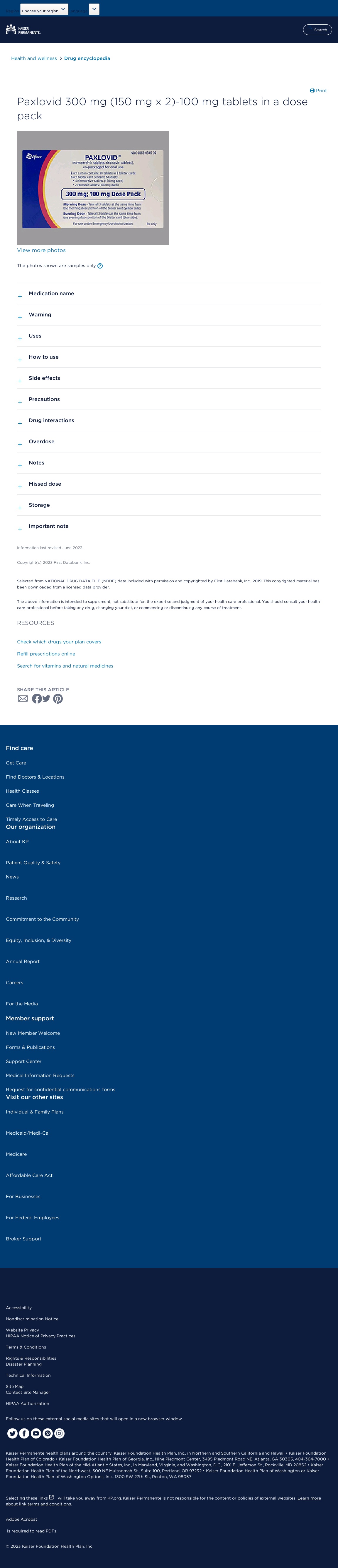
You will take 2 nirmatrelvir tablets and 1 ritonavir pill twice a day for five days.A listing of all medications they're taking, together with over-the-counter medications so the state-licensed pharmacist can display screen for medicine with potentially severe interactions with Paxlovid. Whereas this action permits state-licensed pharmacists to prescribe Paxlovid with certain limitations as described below, group pharmacies not already collaborating as a Take a look at-to-Treat site can resolve if or how they'll provide this service to patients. Immediately, the U.S. Food and Drug Administration revised the Emergency Use Authorization (EUA) for Paxlovid (nirmatrelvir and ritonavir), to authorize state-licensed pharmacists to prescribe Paxlovid to eligible patients, with sure limitations to ensure acceptable affected person assessment and prescribing of Paxlovid. Paxlovid is obtainable under the EUA as tablets of 150 mg of nirmatrelvir co-packaged with a hundred mg tablets of ritonavir; the advisable dose being 2 tablets of nirmatrelvir and one pill of ritonavir twice every day for five days. You will take 2 nirmatrelvir tablets and 1 ritonavir pill twice a day for five days. Other novel aspects embrace prolonged courses of treatment for 10 days and crushing of nirmatrelvir to permit administration by way of an NG tube.
Nonetheless prevention is the most effective medication for any illness.President Biden's administration introduced a collection of actions on Wednesday to make it simpler for states and college districts to supply, recruit, and rent job seeking teachers. When testing optimistic for COVID-19, patients ought to first consider in search of care from their regular health care supplier or locating a Test-to-Treat site of their area. Since she was still testing optimistic months after getting contaminated, Visvabharathy had a hunch-primarily based on her research-that pieces of that virus had been still in her system. Nonetheless prevention is the most effective medication for any illness. The American Academy of Physical Medication and Rehabilitation estimates that 29 million Americans have long Covid. Many patients newly diagnosed with Covid have reported rebound signs after their typical five-day treatment course. An 87-12 months-previous male with a past medical historical past of hypertension and rheumatoid arthritis, not on immune suppression therapy, who has been vaccinated in opposition to COVID-19 was diagnosed with COVID-19 at his nursing home after having signs of shortness of breath, cough, fatigue, and myalgia. A 72-12 months-outdated feminine with a previous medical historical past of rheumatoid arthritis, not on immune suppression therapy, anxiety, and transient ischemic attack, and vaccinated against COVID-19 was diagnosed with COVID-19 after having signs of shortness of breath, cough, and fatigue.
- Sufficient info is not out there to assess for a potential drug interaction
- Modification of different medications is needed on account of a potential drug interplay
- Swelling of the mouth, lips, or face
- Muscle aches
No affected person receiving molnupiravir died of COVID-19. First, the affected person information don't appear to be sufficiently detailed. First, some background. Omicron was initially recognized in Botswana and South Africa in November 2021-although later reports confirmed earlier circumstances in the Netherlands. First, we wonder if patients who have been prescribed Paxlovid were included within the uncovered group (ie, intention-to-deal with design) or did it take 5 full days to be thought-about within the exposed group (ie, per-protocol design)? Most people who take Paxlovid should not experience severe negative effects. While dry mouth isn’t a listed side impact of Paxlovid, it’s a symptom that many people experience. In a clinical trial, Paxlovid decreased the danger of hospitalization and demise by 89% in unvaccinated outpatients with COVID-19 at higher risk of severe disease.(4) Severe adverse events are uncommon with Paxlovid remedy.(4) Paxlovid is given twice day by day for 5 days, beginning as quickly as doable and within 5 days of symptom onset, and is approved to be used in grownup and pediatric patients (12 years of age and older weighing not less than 40kg). Clinicians ought to be aware of the eligibility criteria and the potential for drug interactions with the use of Paxlovid that may preclude Paxlovid use or might require short-term discontinuation of different medications.